VDMN:
PATIENT-FRIENDLY, INJECTION-FREE APPROACH FOR COMMON WARTS
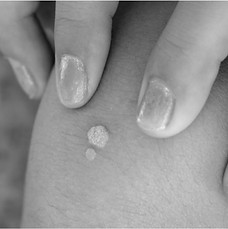
The company’s lead product candidate, VDMN, offers the potential to transform the standard of care for common warts (verruca vulgaris), which affect 19 million Americans and are responsible for millions of clinical visits in the U.S. every year. There are no FDA approved prescription treatments for common warts. VDMN is a novel, injection-free approach to treating common warts in the form of a dissolvable microarray patch incorporating Candida Antigen Extract — which has been successfully used to treat warts for over 20 years with efficacy that exceeds current standard of care. VDMN is painlessly applied to the wart and worn for less than five minutes.
Applications are repeated every three weeks until wart resolution. Because of VDMN’s unique immunologic mechanism of action, patients suffering from multiple warts may only have to treat a single wart to treat all their warts. Because VDMN is a potential first licensure biologic with pediatric labeling, it can expect 12.5 years of regulatory exclusivity after BLA approval. Additionally, VDMN is protected by a broad portfolio of IP that covers the microarray technology and formulation.


VERRUCA VULGARIS
Common Warts
19M
Prevalence (US)
2.2M
annual doctors' visits
1
2
NO FDA-APPROVED PRESCRIPTION TREATMENTS
OTC AND OFF-LABEL TREATMENTS
are either painful, scare children, or require burdensome daily applications for months

INTRALESIONAL IMMUNOTHERAPY
is FDA approved and has been
used successfully off-label for treatment of warts for ~20 years
BUT immunotherapy must be injected into the wart which is painful and requires in office administration
Source: 1. Kwong, P. Prevalence of Common Warts in the United States: Findings From General Population and Physician-Focused Surveys. Annual Fall Clinical Dermatology Conference. 2019, 2. Biotech Value Advisors (BVA) Syndicated Data, Based on Claims Data Analysis, 2021.

Product Drug
Adhesive
Backing
CORE MICROARRAY TECHNOLOGY
Core Microarray Technology Has Been Studied in 300+ Humans including Infants Across 3 Clinical Trials
Tolerability of the Microarray and Efficacy of the Microarray as an Immune Stimulant are Confirmed by All Trials to Date
BIOLOGIC PRODUCT:
CANDIDA ANTIGEN EXTRACT
Candida Antigen’s safety profile is well-characterized as it has been administered to hundreds of thousands of people globally. Primary AEs are Grade 1 Local Skin Reactions